Medical Device Vertical Development
- Home
- /
- Vertical Development
What We Offer
At EEA Innovations, we support medical device vertical development, guiding innovations from early concept through engineering design, prototyping, verification testing, regulatory alignment, and manufacturing readiness. Our integrated approach coordinates engineering, risk management, and compliance activities to help transform promising ideas into regulated, market-ready medical devices efficiently and with reduced development risk.
Our Vertical Development Process:
- Technology Concept Development
- Engineering Design & System Architecture
- Prototype Development
- Verification, Validation & Compliance
- Manufacturing Readiness

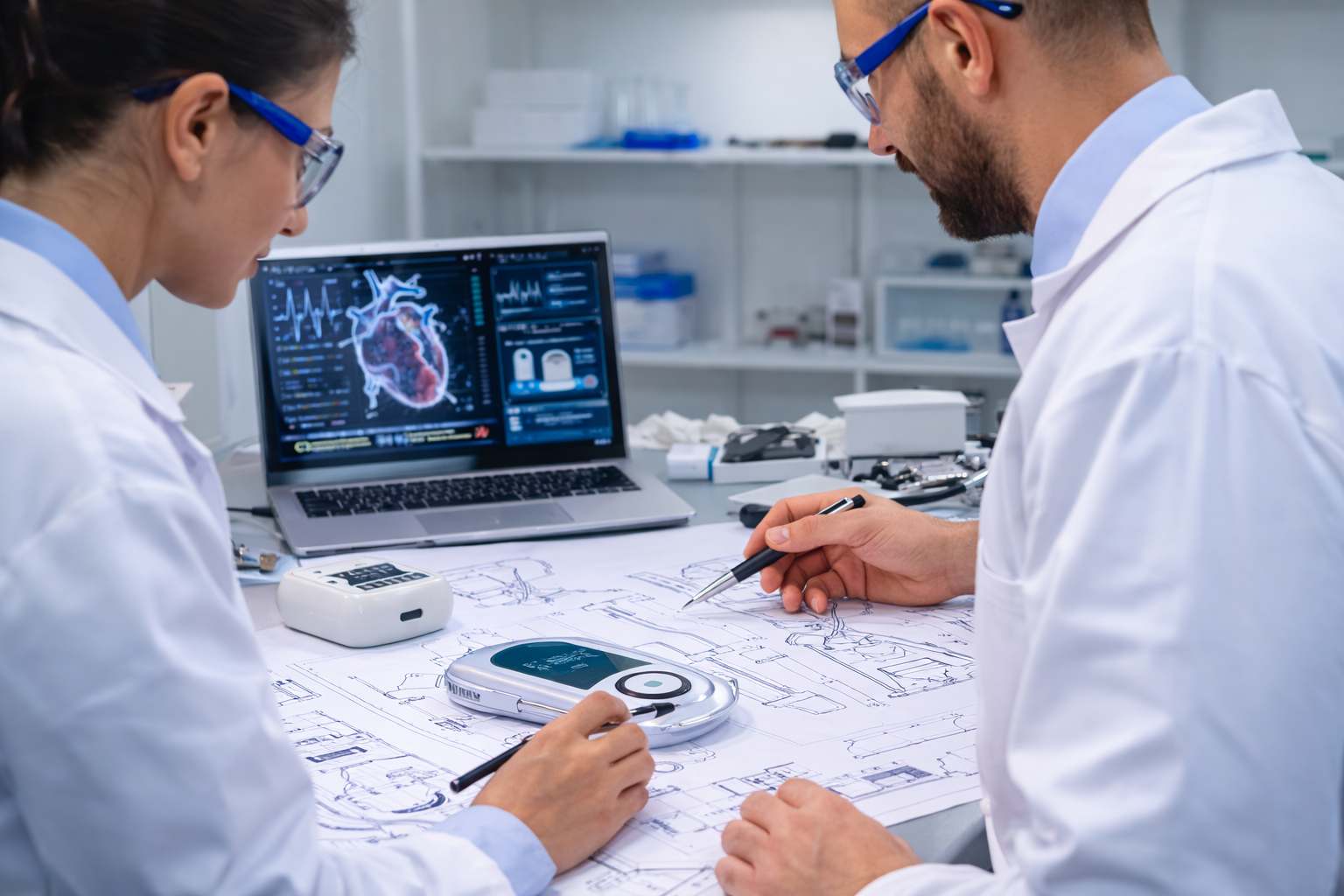
Technology Concept Development
Successful medical device vertical development begins with a strong concept. Our team works closely with innovators and organizations to evaluate technical feasibility, identify potential challenges, and define a clear development strategy. During this phase, we assess the device concept, identify performance requirements, and establish a development roadmap that supports both engineering objectives and regulatory expectations.

Engineering Design & System Architecture
Engineering design is a critical stage in the medical device development process. We develop the system architecture, detailed engineering designs, and technical specifications required to transform concepts into functional medical technologies. Our team ensures that device functionality, usability, safety, and regulatory considerations are integrated from the earliest design stages.

Prototype Development
Prototyping is essential for validating the technical feasibility of a device. Through prototype development, we build functional models that allow teams to evaluate performance, identify design improvements, and refine system capabilities. Iterative prototyping helps reduce development risks and ensures that the device performs as expected before moving to verification and regulatory stages.

Verification, Validation & Compliance
Medical devices must meet strict regulatory and safety requirements. As part of our medical device vertical development services, we support verification and validation activities that confirm the device performs according to design specifications. Our team also ensures alignment with relevant regulatory standards, supporting documentation, risk management activities, and compliance requirements necessary for approval and market entry.

Manufacturing Readiness
As development progresses, the device must be prepared for scalable production. We help organizations transition from development to manufacturing readiness, optimizing design for production, supporting quality system requirements, and preparing the documentation necessary for commercialization. This final stage ensures that the device can be produced reliably while maintaining safety, quality, and regulatory compliance.
